Automated detection of dsRNA contaminants in mRNA samples

Gyrolab® platform runs automated immunoassays in nanoliter-scale and is widely used for pharmacokinetic (PK) studies as an improvement on ELISA and other plate-based assays. The high precision, short assay times and small sample volumes also make it an ideal platform for titer analysis and impurities testing in scarce samples. Double stranded RNA (dsRNA) is one of the main contaminants in synthetic production of mRNA. As dsRNA may cause unwanted immune response it needs to be monitored to ensure the safety of mRNA drugs. In this exploratory work we developed automated Gyrolab assays for quantification of residual dsRNA with limit of detection (LOD) and lower limit of quantification (LLOQ) that match or compete with existing methods.
In vitro transcription (IVT) in mRNA synthesis
The production of mRNA therapeutics for both drug manufacturing and pre-clinical or clinical studies, is dependent on in vitro transcription (IVT). Depending on the IVT conditions byproducts, such as double stranded RNA (dsRNA), might form. These contaminants can trigger unwanted immune response and must therefore be monitored and quantified to ensure the safety and quality of the final mRNA product.
%20in%20mRNA%20synthesis%20blog.png?width=700&height=260&name=In%20vitro%20transcription%20(IVT)%20in%20mRNA%20synthesis%20blog.png)
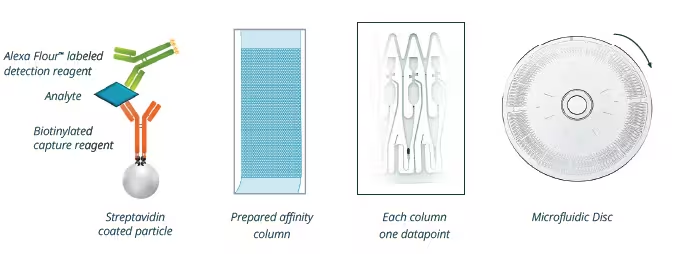
The Gyrolab technology
The Gyrolab platform runs automated immunoassays in nanoliter-scale. Samples and reagents are automatically delivered to 15-nanoliter streptavidin-bead columns on rotating discs by centrifugal and capillary forces. Biotinylated capture reagents bind to the streptavidin-beads and the analytes in samples are detected by Alexa Fluor™ 647 labeled detection reagents. Gyrolab typically requires small sample volumes (a total volume of 5-10 μL for triplicate analysis) and is generally tolerant for different types of biological samples.
Results
Two Gyrolab immunoassays were developed for automated detection of dsRNA contaminants and pg/mL concentrations of dsRNA could be reliably detected, with LOD and LLOQ that match existing methods. The assays were used to analyze mRNA samples, and these preliminary data show promise for Gyrolab as a robust and high throughput method for dsRNA detection.
Labeling and evaluation of antibody pairs
Commercial anti-dsRNA antibodies were labeled with biotin and Alexa fluor™ 647 and tested in different combinations. Three different assay formats were evaluated, standard sandwich assays and double capture or double detect assays for antibody combinations including K2 (IgM) that cannot be labeled directly. Four promising antibody combinations were identified, and the two best ones (Assay 1 and Assay 2) were further optimized.

Two assays with wide dynamic range selected

dsRNA levels analyzed in mRNA samples
In order to further assess the two assays, we measured the dsRNA content in four mRNA samples (samples A-D) with unknown and varying concentrations of dsRNA. Both assays showed reproducible results of dsRNA levels, but assay 2 showed consistently higher concentrations. Example data shown for C and D. Samples with spike recovery <70% and >130% were excluded from the average.


Conclusions
- Automated immunoassays on the Gyrolab system for dsRNA quantification developed and confirmed with quality control (QC) samples
- Two assays with different antibody combinations were successfully used to analyze samples with varying concentrations of dsRNA
- Preliminary results suggest that Gyrolab has potential for reproducible detection of dsRNA contaminants in a high-throughput manner with small sample volumes
Want to know more?